2025 NPS Trends: What Healthcare Providers Need to Know
The illicit drug supply continues to evolve as novel psychoactive substances (NPS) emerge in response to regulatory efforts. Due to constant change in drugs that are circulating, definitive testing methods are required to assist providers in identification of high-risk drug exposure. Findings from Aegis testing data in 2025 highlight ongoing shifts in substance use patterns and underscore the importance of comprehensive, up-to-date toxicology testing to support informed clinical decision making, patient care, and public health response.
Highlights from 2025
Designer opioids were the most frequently ordered NPS class, reflecting continued concern for synthetic opioid exposure and overdose risk.
Xylazine remained the most common compound detected in the NPS‑Other class, despite decreasing in proportion throughout the year, whereas medetomidine represented a substantial and sustained proportion of detections.
The synthetic opioid landscape continues to shift. While fluoro fentanyl remained predominant, other designer opioids—including methylfentanyl analogs and norcarfentanil—demonstrated notable detection alongside emerging and newly identified compounds (e.g. N-propionitrile chlorphine), underscoring the limitations of routine screening panels.
Designer benzodiazepines (DBZDs) remain a significant clinical concern as they are commonly associated with counterfeit prescription pills and may be detected alongside fentanyl reinforcing ongoing risk for overdose. Bromazolam continued to dominate DBZD detections in 2025 despite recent regulatory actions. Alpha-hydroxyphenazolam, added to Aegis testing in June, showed a marked increase in detection during the second half of the year.
Synthetic cannabinoids persist despite regulatory controls. The highly potent cannabinoid MDMB‑4en‑PINACA remained the most prevalent synthetic cannabinoid detected followed by MDMB‑INACA, a precursor of and detected with MDMB-4en-PINACA, both with stable trends observed during the second half of the year.
Substituted cathinones dominated synthetic stimulant detections, with a notable shift in prevalence as N‑isopropyl butylone emerged as the most prevalent synthetic stimulant in 2025.
Most hallucinogen/dissociative detections were attributed to ketamine analogs, particularly 2F-deschloronorketamine and 2F-2-oxo-PCE.
Recommendations for Healthcare Providers
Recognize the limitations of immunoassay (IA)-based testing. Traditional testing panels and IA-only testing approaches are incapable of identifying use of emerging, high-risk substances.
Definitive, mass‑spectrometry–based testing that is routinely updated is necessary for accurate identification of NPS exposure in clinical settings.
Stay informed about emerging drug trends to best understand potential risks to your populations and to improve patient safety and clinical outcomes.
New or Novel Psychoactive Substances (NPS) are a diverse group of synthetic drugs designed to mimic the effects of illicit drugs or prescription drugs used non-medically, while circumventing existing drug control laws.1 NPS may be categorized into various classes including designer opioids, designer benzodiazepines, synthetic cannabinoids, synthetic stimulants, hallucinogens/dissociatives, and others. Since the late 2000’s, the US drug market has experienced a rapidly expanding supply of NPS. In 2024, a record high of 688 individual NPS were reported worldwide to the United Nations Office of Drugs and Crime (UNODC) Early Warning Advisory, including 101 newly identified NPS.2,3
Although not always the case, many NPS are significantly more potent than the drugs they are designed to mimic, which may increase the likelihood of adverse events. NPS often have unknown health risks, and a wide range of adverse effects are associated with use. NPS have been identified in counterfeit prescription pills, increasingly in multi-drug mixtures with unpredictable dosing and compositions.4 As such, NPS have been associated with numerous overdose outbreaks and reports of death.5
In response to increasing public health concerns, legislative and regulatory actions have largely targeted specific compounds, chemical structures or classes of NPS.6 However, the synthetic drug market has continued to adapt and expand with new classes of NPS emerging; and “second” and “third generation” compounds appearing in the illicit drug supply from NPS classes already regulated. Once an NPS has been deemed a controlled substance, new or chemically modified, non-regulated NPS often appear. Not all NPS are new; some are compounds previously developed for potential use as a pharmaceutical drug but never brought to market. Others are veterinary medications or approved medications in other countries and are classified as an NPS when first identified in the illicit drug supply. NPS may change frequently as legislation to control specific chemical structures or classes of NPS is introduced, creating a cat-and-mouse dynamic between regulators and illicit manufacturers, resulting in continual evolution of NPS in the illicit drug supply. This remains a challenge for regulatory and enforcement agencies, monitoring institutions, clinical and toxicology laboratories, as well as healthcare providers.
Healthcare professionals should be aware of emerging drugs and new trends in drug use as they can inform individual patient care decisions as well as public health and safety response. Frequent NPS use has been identified in individuals with substance use disorders and mental health diagnoses.7-12 NPS use can have a negative impact on mental health outcomes and undermine treatment goals and effectiveness.13,14 Furthermore, NPS use can go undetected when only traditional drug testing panels are utilized or when clinical decisions rely on immunoassay (IA) or point of care testing (POCT). IA-based testing relies on cross-reactivity to a target drug; however, the extent of cross-reactivity varies across drugs within a class. Lack of cross-reactivity across a class may result in false negatives. There are multiple guidelines recommending definitive testing following unexpected POCT results. 15,16 IA tests cannot identify specific NPS because this methodology lacks specificity. If IA is utilized for rapid initial testing, then providers evaluating results should consider follow-up testing using definitive testing methods such as mass spectrometry to confirm the presence of a particular NPS. Additionally, the use of IA for NPS testing is complicated by the rapid change of the NPS market and is not ideal and may be prone to false negative results.
To appropriately test for NPS, definitive testing methods should be utilized and need to be frequently updated to account for emerging NPS. Aegis is committed to this effort and updated its NPS testing menu in June and December of 2025. Aegis offers NPS testing by class in both urine and oral fluid using definitive testing by mass spectrometry. Test offerings include designer opioids, designer benzodiazepines, synthetic cannabinoids, synthetic stimulants, hallucinogens/dissociatives, and NPS-Other.
The goal of this clinical update is to review the prevalence of NPS detected at Aegis in the second half of 2025 compared to annual data. For additional information, please also see our 2025 Mid-year Update on NPS Trends.
Figure 1 shows NPS order rates by class for 2025. Among samples with any NPS class ordered, designer opioids were the most frequently ordered throughout the year. Order rates for the remaining NPS classes were lower and remained relatively stable over time. When evaluating NPS data, it is important to consider that differences in order rates may impact observed NPS detection and prevalence between classes.
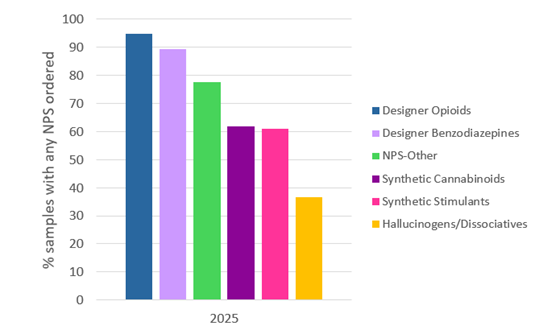
Figure 1. NPS order rate by class as % of samples with any NPS class ordered in 2025
NPS-OTHER
The NPS-Other category includes substances that do not easily fit within other designated NPS classifications. Not all compounds in this class are illicitly-manufactured; some are veterinary medications or other substances that have been identified as adulterants in the illicit drug supply. For example, xylazine is an alpha-2 adrenergic receptor agonist approved for veterinary use as a sedative with analgesic and muscle relaxant properties. However, it is not approved for use in humans and may cause unique withdrawal episodes and severe necrotic skin ulcers. The United States (U.S.) Food and Drug Administration (FDA) previously released an alert regarding risks to patients exposed to xylazine in the illicit drug supply.17 Figure 2 shows the proportion of compounds detected in the NPS-Other class in Q3, Q4 and annually in 2025; quarterly trends for the first half of 2025 are described in the 2025 Mid-year Update on NPS trends. In Q1, xylazine and its metabolite represented approximately 70% of detections in the NPS-Other class and its proportion decreased in Q2. In Q3, xylazine and its metabolite represented approximately 61% of detections in the NPS-Other class and decreased to approximately 54% in Q4. Annually, xylazine and its metabolite made up 63% of compounds detected in the NPS-Other class.
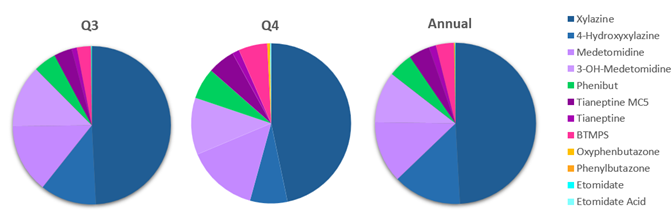
Figure 2. Proportion of compounds detected in the NPS-Other class in Q3, Q4 and Annually in 2025
Medetomidine and its metabolite 3-hydroxy medetomidine were the next most prevalent compounds in the NPS-Other class. Like xylazine, medetomidine is an alpha-2 adrenergic receptor agonist and has been detected as an adulterant in the illicit drug supply.18 Recently, medetomidine has been reported to be rapidly proliferating across the U.S.19 The proportion of medetomidine and its metabolite increased from Q1 to Q2 but remained similar in Q3 and Q4 (~26-27%). Annually, medetomidine and its metabolite were approximately 22% of detections in the NPS-Other class.
Phenibut was the next most prevalent compound in the NPS-Other class accounting for ~5% of annual detections. Phenibut is a synthetic gamma aminobutyric acid (GABA) analog with benzodiazepine-like effects, marketed in the U.S. as anti-anxiety, nootropic or sleep aid supplements. However, FDA has declared that phenibut is not a legitimate dietary ingredient and has issued warning letters to companies selling products containing phenibut.20 Use of phenibut has been associated with adverse effects and can lead to dependence and withdrawal upon cessation of use.21 The proportion of phenibut detections increased slightly from Q3 to Q4. Like phenibut, tianeptine and its metabolite tianeptine MC5 accounted for ~5% of the detections in the NPS-Other class in 2025. Tianeptine, also called “gas station heroin”, is structurally classified as an atypical tricyclic antidepressant but at high doses it has mu opioid receptor activity. Thus, misuse can result in opioid-like highs, and users can develop tolerance and dependence. The FDA reported that tianeptine products have been linked to serious adverse events including overdose and death.22,23
BTMPS is a potent Ca2+ channel blocker and industrial chemical used in the manufacture of plastics.24 It rapidly appeared in the illicit drug supply in summer of 2024 and was added to Aegis testing in December 2024. BTMPS has since been identified as a public health concern. 25,26 BTMPS represented ~4% of detections in the NPS-Other class in 2025, with a notable increase from ~3% in Q3 to ~6% in Q4.
Phenylbutazone, a nonsteroidal anti-inflammatory drug withdrawn from the market due to severe adverse effects, and its metabolite oxyphenbutazone were added to Aegis NPS-Other testing in December of 2024. Etomidate, a sedative-hypnotic used in critical care and emergency medicine, was added in June 2025, and its metabolite etomidate acid was included in December 2025. Both have been detected in the illicit drug supply, but together accounted for less than 1% of NPS-Other positives in 2025.27-29
Designer Opioids
Designer opioids are comprised of multiple subclasses including fentanyl analogs, “nitazene analogs,” and others. Figure 3 shows the proportion of designer opioids detected in Q3, Q4 and annually in 2025; quarterly trends for the first half of 2025 are described in the 2025 Mid-year Update on NPS trends. The fentanyl analog fluoro fentanyl has three positional isomers (meta-, ortho- and para-) that are not distinguished in Aegis’ designer opioids test. The para-fluorofentanyl isomer specifically has been associated with overdose deaths.30,31 For multiple years, fluoro fentanyl was the most frequently detected NPS at Aegis among all NPS classes. However, in 2023 xylazine overtook it as the most prevalent NPS detected at Aegis irrespective of NPS class. Since then, fluoro fentanyl has remained the second most prevalent NPS detected at Aegis. Fluoro fentanyl and related compounds, despropionyl fluorofentanyl and fluoro norfentanyl, have been the most prevalent designer opioids detected at Aegis for several years. In 2022 para-fluorofentanyl specifically was detected in 67 (44.7%) of 310 counterfeit oxycodone pills.4 More recently a decrease in the prevalence of fluoro fentanyl and related compounds has been observed. For example, in Q3 these compounds represented ~54% of the designer opioid detections, whereas in Q4 that percentage dropped to 48%, but annually they represented 56% of the 2025 designer opioid detections.
Methylfentanyl related compounds made up the second and third most prevalent designer opioids detected in 2025. Methylfentanyl has three positional isomers (ortho-, meta- and para-) and was added to Aegis’ designer opioid testing in December of 2024. In Aegis’ testing, the ortho- isomer is distinguished from the meta- and para- isomers, which are not distinguished from one another. O-methylfentanyl and its metabolite despropionyl-o-MF together were the second most detected designer opioids in 2025, and despropionyl m/p-methylfentanyl (metabolite of m/p-methylfentanyl) was third most detected. The same month methylfentanyl testing was implemented at Aegis, the Center for Forensic Science Research and Education (CFSRE) released a public alert noting that ortho-methylfentanyl, the newest synthetic opioid identified in fatal drug overdoses, was proliferating across North America.32 In 2025, o-methylfentanyl and its metabolite represented 15% of designer opioid detections in Q3 and 16% of detections in Q4, but 13% annually. For m/p-methylfentanyl, the percent change in proportion between Q1 and Q2 more than doubled (data previously reported), annually it made up just under 8% of designer opioid detections, which was similar to but slightly less than its Q3 proportion. In Q4, m/p-methylfentanyl represented ~10% of designer opioid positive results. Both o-methylfentanyl and m/p-methylfentanyl had a greater proportion of detections in Q3 and Q4 than when considered annually.
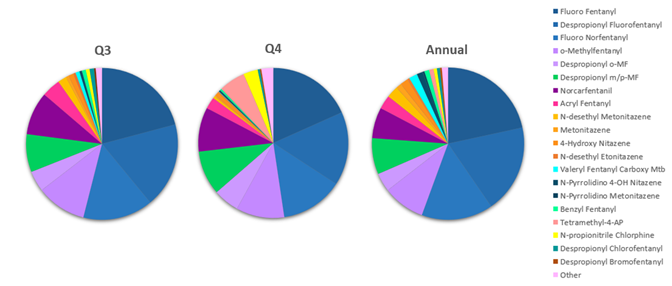
Figure 3. Proportion of Designer Opioids detected in Q3, Q4 and Annually in 2025. Designer Opioids with less than 50 detections in 2025 were combined and represented as “Other”.
Norcarfentanil, metabolite of carfentanil and remifentanil, was the next most prevalent designer opioid in 2025. Carfentanil is a veterinary sedative for large animals and not approved for use in humans. It is estimated to be one hundred times more potent than fentanyl and has been found in counterfeit prescription pills. The United States Drug Enforcement Administration (DEA) reported an increased presence of carfentanil in the illicit drug supply and noted its association with overdose deaths. Comparing January-June of 2023 to the same period in 2024, carfentanil involved overdose deaths increased seven-fold.33 Like m/p-methylfentanyl, the percent change in proportion of carfentanil detections more than doubled from Q1 to Q2 (data previously reported). Additionally, its proportions in Q3 and Q4 were greater than when considered annually. The percent proportion in Q3 and Q4 was similar at ~9-10% of detections whereas annually it made up roughly 6.5% of designer opioid positives.
Nitazene and N-pyrrolidino nitazene analogs were some of the designer opioids that saw significant decreases in proportion of designer opioids detected between Q1 and Q2 (data not shown). This decrease in proportion of nitazene and N-pyrrolidino nitazene analogs continued in the second half of the year. For example, metonitazene and its N-desethyl metabolite made up ~3.7% of designer opioid detections in 2025 but only ~0.7% in Q4. Similarly, N-pyrrolidino metonitazene and generic shared metabolite N-pyrrolidino 4-OH nitazene detections annually were ~1.7% of designer opioid detections, whereas in Q4 they represented ~0.4%. Nitazenes began to appear in the U.S. illicit drug supply in 2019 following legislation targeting fentanyl-related substances. The rise in nitazene analogs in the illicit drug supply quickly became a public health concern as nitazene-related overdose deaths were reported.34 Many nitazenes have similar or greater potency than fentanyl with some even significantly more potent than fentanyl and thus they have increased risk of overdose and mortality. Initiatives to control nitazenes have been implemented or introduced, for example in the summer of 2025, the DEA temporarily scheduled some nitazene analogs including N-Pyrrolidino metonitazene, N-desethyletonitazene and N-desethylprotonitazene. Additionally, the 119th Congress has introduced the Nitazene Control Act, which is legislation to control nitazenes as a class. Thus, these efforts may be impacting the decrease in proportion of nitazene analogs detected in 2025.
Designer opioids with less than 50 detections in 2025 were included in the “Other” count. In December of 2025, three new compounds were added to Aegis’ designer opioid testing, one of which exceeded 50 detections annually, despite their addition nearing mid-month of the last month of the year. CFSRE issued a public alert in May of 2025 regarding the discovery of tetramethylfentanyl (TMF)-related substances including tetramethyl-4AP and tetramethyl norfentanyl and the presence of BTMPS in the illicit drug supply.35 Despite only being included in testing for a little more than half a month, tetramethyl-4AP detections made up 5.7% of designer opioid detections in Q4, and a little less than 1% of detections in 2025.
N-propionitrile chlorphine, also known as cychlorphine, was added to Aegis testing in June of 2025 and showed a notable increase from <1% of detections in Q3 to 3.1% in Q4. This compound belongs to an emerging class of designer opioids identified as “orphine” analogs. In vitro data indicates N-propionitrile chlorphine is approximately ten times more potent than fentanyl.36 N-propionitrile chlorphine was first identified by CFSRE mid-2024. However, detection increased in late 2025, and early 2026, and it has recently been associated with a number of overdose deaths, including at least 16 in Tennessee.36,37 CFSRE reported detection of N-propionitrile chlorphine in 25 fatal overdose cases, and noted that it was the sole opioid present in 11 of 25 cases.36 However like most NPS, it has been detected with other NPS. It has been detected in cases from all regions of the U.S. and in Canada.36 It is important to note that N-propionitrile chlorphine is not detected with standard screening or POCT and requires specialty testing and few laboratories are currently testing for it.
Designer Benzodiazepines
Designer benzodiazepines (DBZDs) are a major public health concern, with their prevalence in the illicit drug supply increasing substantially over the past decade.38 These substances are frequently identified in counterfeit prescription medications, such as fake “Xanax” pills or other alprazolam medications that may appear indistinguishable from the legitimate pharmaceutical medication. In addition to containing DBZDs, counterfeit pills may contain other substances including fentanyl.39 As central nervous system (CNS) depressants, DBZD may produce serious adverse effects and drug-drug interactions, particularly when used in combination with other benzodiazepines or CNS depressants.
Data from Centers for Disease Control and Prevention (CDC) highlight public health risks associated with counterfeit pill use. Evidence of counterfeit pill use was associated with higher percentages of overdose deaths involving illicitly manufactured fentanyl (93.0% versus 72.2%) and illicit DBZDs (5.3% versus 1.4%) compared to deaths without evidence of counterfeit pill use. These findings underscore the importance of overdose prevention messaging that emphasizes the dangers associated with counterfeit prescription pills.
A prior evaluation of internal Aegis data demonstrated that among samples in which a DBZD was detected, there was a 38% co-positivity with traditional benzodiazepines,40 indicating that DBZD use may occur alongside prescribed benzodiazepines. In one internal study, a point-of-care testing (POCT)-positive sample, consistent with prescribed benzodiazepine use, had two DBZDs present in addition to the prescribed benzodiazepine. In contrast, another sample that was POCT-negative contained four DBZDs.41 These findings indicate that healthcare providers who rely solely on POCT results to assess compliance with prescribed benzodiazepines may fail to detect illicit DBZD use. Additionally, patients who obtain what appear to be legitimate pharmaceutical medications outside of licensed pharmacies may unknowingly be exposed to counterfeit medications containing DBZDs, fentanyl, or other synthetic opioids.
Detection of DBZDs presents additional challenges due to limitations of immunoassay-based screening. Designer drugs often have modified chemical structures, which can result in both false-negative and false-positive immunoassay results. In a study evaluating thirteen DBZDs across multiple immunoassay platforms, most DBZDs demonstrated high cross-reactivity; however the DBZD with the greatest structural divergence compared to other benzodiazepines, had the lowest detectability.42 Consequently, DBZDs may not be reliably detected when their chemical structure differs substantially from the assay’s target compound. In an internal study, samples were evaluated using two separate POCT platforms and definitive testing. Thirty-five of forty-eight samples had negative (32) or discrepant (3) POCT results, among these, 30 samples had only DBZDs present after definitive testing, four containing multiple DBZDs.41 Moreover, immunoassays lack specificity and cannot identify which benzodiazepines are present, how many are present, or whether a positive result reflects true benzodiazepine exposure versus cross-reactivity with unrelated compounds.
Regulatory actions have attempted to address the proliferation of DBZDs. Figure 4 shows the proportion of designer benzodiazepines detected at Aegis in Q3, Q4, and annually in 2025; quarterly trends for the first half of 2025 are described in the 2025 Mid-year Update on NPS trends.
In July 2023, the DEA temporarily placed clonazolam, etizolam, flualprazolam, flubromazolam and diclazepam in Schedule I of the Controlled Substances Act (CSA) to limit access to these substances.43 At that time bromazolam remained unscheduled and became the most prevalent DBZD detected at Aegis in 2023. Bromazolam has since remained the most prevalent DBZD based on annual positivity.
Throughout 2024 and 2025, several states implemented scheduling of bromazolam, and in December 2025, the DEA issued notice of its temporary placement in Schedule I of the CSA.44 In 2025, bromazolam and its metabolite alpha-hydroxybromazolam, accounted for 68% of annual DBZD detections at Aegis; however, this proportion declined from 64% in Q3 to 55% in Q4. Similarly, the detection of 8-aminoclonazolam, which was the second most prevalent DBZD annually, was significantly less in Q3 and Q4 than in Q1, when it represented nearly 20% of DBZD detections.
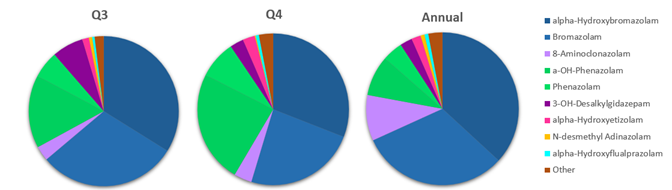
Figure 4. Proportion of Designer Benzodiazepines detected in Q3, Q4 and Annually in 2025. Designer Benzodiazepines with less than 50 detections in 2025 were combined and represented as “Other”.
Notably, phenzolam and its metabolite, alpha-hydroxyphenazolam (added to Aegis testing in June of 2025), showed a marked increase in the proportion of detections in the second half of the year, increasing from 22% of DBZD detections in Q3 to 32% in Q4, while representing 13% annually.
The next most prevalent DBZD was desalkylgidazepam. Its metabolite, 3-hydroxydesalkylgidazepam, was added to Aegis testing in June 2025 and appeared to improve detection of desalkylgidazepam use. In Q3, 3-hydroxydesalkylgidazepam accounted for ~7% of DBZD detections but decreased by more than half to 3% in Q4, remaining at 3% annually. Alpha-hydroxyetizolam, increased modestly from Q3 to Q4 and was the next most prevalent DBZD, accounting for 2% of detections annually. N-desmethyl adinazolam whose proportion decreased substantially from Q3 to Q4 and alpha-hydroxyflualprazolam each represented less than 1% of detections in Q3, Q4 and annually. DBZDs with fewer than 50 annual detections were grouped and designated as “Other” and collectively accounted for 3% annual DBZD detections.
Synthetic Cannabinoids
Synthetic cannabinoids were among the first NPS to appear in the US in the late 2000s. Although commonly referred to as “K2” or “Spice,” these substances are sold under numerous other names and are widely available through convenience stores, smoke or tobacco shops, and online marketplaces. Synthetic cannabinoids are typically sprayed onto or mixed with plant material and marketed as “legal highs” or herbal incense products, often labeled “not for human consumption.”
Synthetic cannabinoids are compounds designed to selectively bind to cannabinoid receptors. Many were originally developed as potential therapeutic agents, whereas others were synthesized for illicit use to mimic the effects of delta‑9‑tetrahydrocannabinol (THC), the primary psychoactive component of Cannabis sativa. Unlike THC, however, synthetic cannabinoids are often full agonists at cannabinoid receptors and exhibit high binding affinity, making them significantly more potent than THC.
In recent years, the DEA has reported a decreasing number of submissions of synthetic cannabinoids to DEA forensic laboratories.45 This decline may be attributable, in part, to legislation targeting specific chemical structures and classes of NPS. In 2021, China, often identified as a source of synthetic drugs, implemented a class‑wide ban on synthetic cannabinoid receptor agonists (SCRAs), covering seven common SCRA generic structures. In response to these regulatory efforts, multiple structural modifications to SCRAs have emerged, including for example “tail‑less” analogs such as ADB‑INACA.⁴6
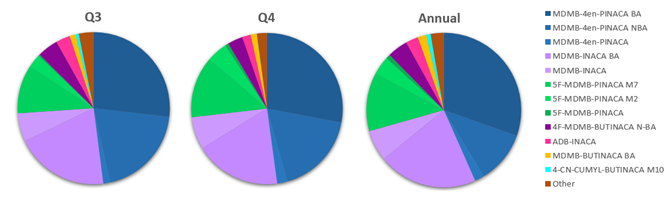
Figure 5. Proportion of Synthetic Cannabinoids detected in Q3, Q4 and Annually in 2025. Synthetic Cannabinoids with less than 50 detections in 2025 were combined and represented as “Other”.
Figure 5 shows the proportion of synthetic cannabinoids detected at Aegis in Q3, Q4, and annually in 2025; quarterly trends for the first half of 2025 are described in the 2025 Mid-year Update on NPS trends.
MDMB‑4en‑PINACA has been the most prevalent synthetic cannabinoid detected at Aegis since 2022. Detection occurred predominantly via its butanoic acid (BA) metabolite and, to a lesser extent, its N‑BA metabolite, which was added to Aegis testing in June 2025. In December 2023, MDMB‑4en‑PINACA was temporarily placed in Schedule I of the CSA.47 Yet, MDMB-4en-PINACA and related compounds increased in proportion from Q1 to Q2, then remained the stable in Q3 and Q4, accounting for 48% of the synthetic cannabinoid detections in both quarters. Annually, MDMB‑4en‑PINACA and metabolites represented 43% of detections and it remained the most prevalent synthetic cannabinoid detected at Aegis.
MDMB-INACA is a synthetic cannabinoid precursor that can be used to make several different synthetic cannabinoids including MDMB-4en PINACA.48 Detected primarily via its BA metabolite, MDMB‑INACA was frequently identified alongside MDMB‑4en‑PINACA and was the second most prevalent synthetic cannabinoid detected in 2024, a pattern that continued in 2025. In Q1 of 2025, MDMB-INACA and its BA metabolite together accounted for nearly one‑third of synthetic cannabinoid detections. In Q3 and Q4, this proportion declined modestly to ~25–26%. Annually, MDMB‑INACA and its metabolites represented 28% of detections, making them the second most prevalent synthetic cannabinoids in 2025.
5F‑MDMB‑PINACA, detected via its M7 and M2 metabolites, represented ~20% of positive results in Q1 2025 but declined to about 13% of detections in Q3. However, the proportion increased again in Q4 to 18%, resulting in an annual proportion of 17% and ranking 5F‑MDMB‑PINACA metabolites as the third most prevalent synthetic cannabinoids detected in 2025.
Detections of 4F‑MDMB‑BUTINACA N‑BA metabolite, ADB‑INACA, and MDMB‑BUTINACA BA metabolite in Q3 were similar to their annual proportions and declined only slightly in Q4. In contrast, detection of 4‑CN‑CUMYL‑BUTINACA M10 was highest in Q1 and declined throughout the year, with detections in the second half of 2025 accounting for less than 1% of results. Synthetic cannabinoids with fewer than 50 annual detections were grouped into the “Other” category and collectively represented ~3% of annual synthetic cannabinoid detections.
Synthetic Stimulants
Shortly after the emergence of synthetic cannabinoids, synthetic cathinones began appearing in the U.S. drug market. These substances are commonly marketed as “bath salts” or “plant food” and are typically labeled “not for human consumption” to evade regulatory oversight. As regulatory efforts expanded, multiple subclasses of synthetic stimulants emerged but synthetic cathinones continue to represent the most prevalent type of synthetic stimulant in the illicit drug supply.
Synthetic cathinones are chemically related to cathinone, a naturally occurring psychoactive constituent of the khat plant. Their pharmacologic effects resemble those of methamphetamine, cocaine, and MDMA, and they are frequently sold as MDMA or “Molly.” Desired effects may include euphoria, increased energy and alertness, empathy, talkativeness, enhanced sociability, and perceived sexual performance. Reported adverse effects include hallucinations, agitation, excited delirium, paranoia, panic attacks, tachycardia, hypertension, severe psychosis, hyperthermia, suicidal ideation or behaviors, coma, and death.
Although traditional toxicology testing for synthetic cathinones is widely available, maintaining clinical relevance requires awareness of which compounds are currently circulating in the illicit drug supply. Testing panels that are not routinely updated may target compounds that are no longer prevalent in the illicit drug supply, limiting their clinical utility. Ongoing updates to synthetic stimulant testing panels are therefore essential for accurate detection and identification of the use of synthetic stimulant drugs.
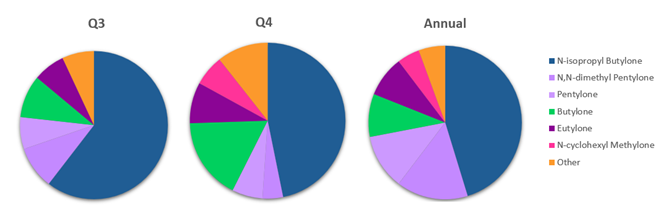
Figure 6. Proportion of Synthetic Stimulants detected in Q3, Q4 and Annually in 2025. Synthetic Stimulants with less than 10 detections in 2025 were combined and represented as “Other”.
Figure 6 shows the proportion of synthetic stimulants detected at Aegis in Q3, Q4, and annually in 2025; quarterly trends for the first half of 2025 are described in the 2025 Mid-year Update on NPS trends.
N,N-dimethylpentylone and its metabolite pentylone have been the most prevalent synthetic stimulants detected at Aegis since 2022. However, following decreasing detection throughout 2024, they continued to decrease in 2025, declining from 50% of detections in Q1 to ~11% in Q4. Annually, N,N-dimethylpentylone and pentylone accounted for ~27% of detections ranking second among synthetic stimulants detected in 2025.
N-isopropyl butylone, a novel substituted cathinone first identified by CFSRE in August 2024 and added to Aegis Synthetic Stimulant testing in December 2024,49 emerged as the most prevalent synthetic stimulant detected at Aegis in 2025. In Q1, N-isopropyl butylone represented ~27% of synthetic stimulant detections and nearly doubled in proportion from Q1 to Q2. Its highest prevalence occurred in Q3 at ~61% followed by a decrease to ~47% in Q4. Annually, N-isopropyl butylone accounted for 45% of synthetic stimulant detections and was the most prevalent synthetic stimulant detected at Aegis.
The largest increase in detections during the first half of 2025 was observed for butylone, which rose from 2.6% of detections in Q1 to nearly 11% in Q2. Its proportion was ~9% in Q3 and increased further to 17% in Q4, resulting in an annual proportion of 9% and ranking it as the third most prevalent synthetic stimulant. This was comparable to eutylone, which accounted for just under 9% of detections annually, despite ranking third in Q1 at ~10%. Notably, all butylone positive samples in 2025 were copositive for N-isopropyl butylone.
Synthetic stimulants with fewer than 10 detections in 2025 were grouped into the “Other” category. The proportion of these compounds increased from 7% in Q3 to nearly 11% in Q4 and represented ~6% of all synthetic stimulant detections annually.
Hallucinogen/Dissociatives
Hallucinogens/Dissociatives class of NPS comprises compounds designed to mimic drugs like lysergic acid (LSD), phencyclidine (PCP) and ketamine, which act at serotonin (5-HT2A) and/or N-methyl-D-aspartate (NMDA) receptors to produce hallucinations or anesthetic and dissociative effects. The proportion of hallucinogens/dissociatives detected in Q3, Q4, and annually in 2025 is shown in Figure 7; trends observed during the first half of 2025 are described in the 2025 Midyear Update on NPS Trends.
The ketamine analog 2Fdeschloroketamine and its metabolite, 2F-deschloronorketamine, first appeared in April 2022. 2F-deschloronorketamine became the most prevalent hallucinogen/dissociative detected in 2022 and remained so through 2025. In Q1, it accounted for ~58% of hallucinogens/dissociatives detected, increasing to 62% in Q3 and 72% in Q4. Annually, it represented 63% of detections within this class.
The next most prevalent compound was 2F-2-oxo-PCE, which accounted for ~34% of detections annually. 2F-2-oxo-PCE is a positional isomer of fluorexetamine and is structurally similar to ketamine. Although ketamine is a federally scheduled substance, 2F-2-oxo-PCE is not currently scheduled at the federal level; however, some states have classified it as a controlled substance. 2F-2-oxo-PCE was added to Aegis NPS testing in September 2023. In May 2024, CFSRE released a public alert reporting detection of 2F-2-oxo-PCE in recreational drug markets across North America, noting that more than 60% of samples also contained opioids, primarily fentanyl.50
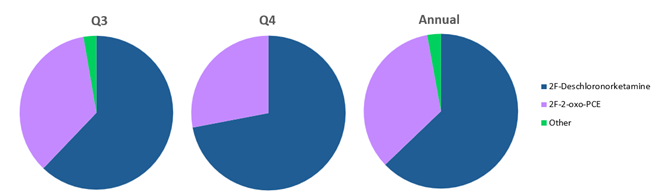
Figure 7. Proportion of Hallucinogens/Dissociatives detected in Q3, Q4 and Annually in 2025. Hallucinogens/Dissociatives with less than 5 detections in 2025 were combined and represented as “Other”.
All NPS
The Top NPS detected at Aegis in the first half of 2025 and annually are shown in Table 1. The two most prevalent NPS in 2025, irrespective of NPS class, were xylazine and fluoro fentanyl (and related compounds), which were also the top two NPS in 2024. Overall, the relative ranking of top NPS remained largely consistent from Q1–Q2 through the end of the year, with three notable exceptions: medetomidine increased from the fifth most prevalent to the third most prevalent, norcarfentanil rose three positions, and the designer benzodiazepine 8-aminoclonazolam declined by five positions.
Several compounds introduced in December 2024 appeared on the top NPS list even within the first half of 2025, including the designer opioids o-methylfentanyl and m/p-methylfentanyl (detected as despropionyl metabolite), as well as the emerging adulterant BTMPS. Additionally, the inclusion of the phenazolam metabolite alpha-hydroxyphenazolam in June 2025 improved detection of phenazolam, contributing to its appearance among the top NPS detected in 2025.
NPS-Other was the most represented NPS class in the top NPS detected followed by designer opioids and then by designer benzodiazepines and synthetic cannabinoids. Differences in NPS class order rates may influence these findings, as ranking in Table 1 is based on the total number of detections.
Table 1. Top NPS Detected at Aegis Q1-Q2 and 2025
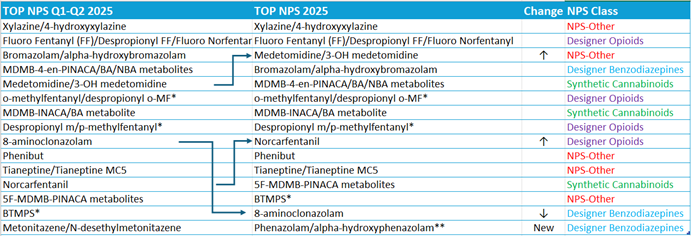
*added in Dec 2024, **added in Jun 2025
Please call our clinical scientists at 1-877-552-3232 if you require additional information.
NOTICE: The information above is intended as a resource for health care providers. Providers should use their independent medical judgment based on the clinical needs of the patient when making determinations of who to test, what medications to test, testing frequency, and the type of testing to conduct.


